Current Projects
LIMBOW: Linking Ice Melt with the Biogeochemistry and Organisms of the Weddell Sea
Sea ice is a key feature of the Southern Ocean that shapes the physical structure of the water column and regulates phytoplankton community dynamics and primary production. Phytoplankton are the base of the food chain, and the type of phytoplankton present, along with their overall productivity, impact the abundance of zooplankton and larger animals. Phytoplankton communities and production are also an important link for carbon export to the deep sea, a critical service provided by the Southern Ocean. However, sea ice extent and duration is decreasing in the Antarctic Peninsula region of the Southern Ocean, potentially affecting carbon export. This project aims to evaluate physical and chemical characteristics of sea ice in the Weddell Sea and quantify effects of melting sea ice on phytoplankton and zooplankton growth and export.
Our interdisciplinary team of physical, chemical, and biological oceanographers will comprehensively evaluate characteristics of sea ice in the western Weddell Sea, and parse the effects of sea ice melt on biological systems and carbon export through a multi-pronged approach involving environmental observations and replicated factorial incubation experiments. Snow pits and ice cores will be used to characterize physical (snow density, hardness, temperature, and grain size and shape) and chemical parameters (δ¹⁸O, nitrate + nitrite and Fe concentrations) of sea ice. Physical characteristics will be used to better calibrate satellite observations and improve future remote sensing. Chemical parameters will help determine how much meltwater is sea-ice derived and will further constrain the impact of sea ice melt on macro and micro nutrient availability in the surface mixed layer. The spatial evolution of sea ice melt across the Weddell Sea continental shelf will be quantified through aerial imagery and ocean observing transects extending away from the marginal ice zone. These transects will include seawater sampling, zooplankton tows, and deployment of an in situ particle imager to characterize microbial, phytoplankton, and zooplankton communities and calculate particle flux. Experimental incubations will constrain the impacts of changing salinity, light, and nutrient regimes associated with ice melt on biological productivity, particle formation, and export potential. Overall, this work will enable better predictions of the ecosystem effects from changing sea ice conditions, such as shifts in krill abundance and its ability to support macrofauna and fisheries, and changes in carbon export. This project is supported by NSF OPP Award 2532479.
 From left: The team in Punta Arenas before setting sail for the Weddell Sea; The Sikuliaq in the ice, drone photo from Co-PI Ellen Buckley; The team in the Neumayer Channel; Maggi, Ellen, and MTs sampling a sea ice pond.
From left: The team in Punta Arenas before setting sail for the Weddell Sea; The Sikuliaq in the ice, drone photo from Co-PI Ellen Buckley; The team in the Neumayer Channel; Maggi, Ellen, and MTs sampling a sea ice pond.
B-vitamins, bacteria, and harmful algal blooms (HABs)
The dinoflagellate Pyrodinium bahamense is found in subtropical and tropical estuaries globally and biosynthesizes the neurotoxin Saxitoxin. Saxitoxin can bioaccumulate in filter-feeding shellfish and other marine organisms. If these contaminated food sources are consumed by humans, Paralytic Shellfish poisoning may ensue, which can lead to death. As a result, Pyrodinium bahamense blooms are a major public health concern, and disentangling the many factors that contribute to Pyrodinium bloom frequency and intensity is a critical research area. While many HAB-forming dinoflagellates are known to require external sources of B-vitamins to grow, the B-vitamin requirements for Pyrodinium had not been reported. B-vitamins can be limiting in coastal and open ocean regions and could impact bloom formation. MICOlab master’s student, Lydia Ruggles Howe, showed that Pyrodinium is a vitamin B12 (cobalamin) auxotroph (i.e., it cannot grow without a B12 source). She also found that bacteria Isolated from Indian River Lagoon (FL) when the was an active P. bahamense bloom could partially restore P. bahamense growth in culteres where B12 was witheld. In contrast, bacteria isolated from Old Tampa Bay (FL) did not restore growth when B12 was witheld. Thus, bacteria in seawater can fulfill at least some of P. bahamense’s B12 requirement, but other anthropogenic sources may supplement bacterial support bloom-level growth. This research was supported in part by a Phycological Society of America (PSA) grant-in-aid of research and the Sanibel-Captiva Shell Club grant.
Related Publication: Ruggles Howe, L., D’Amore, L., Lopez, C., Shankar, S., Mars Brisbin, M. (2025) B-vitamin requirements and phycosphere interactions for the HAB-forming dinoflagellate, Pyrodinium bahamense var. bahamense. bioRxiv. link.
To follow this up, we are investigating seasonal and spatial patterns in vitamin B12 availability throughtout Old Tampa Bay, while simultaneously measuring phytoplankton and bacteria abundance and activity. Since different forms of the B12 molecule come from different sources (e.g., cyanocobalamin is the synthetic form found in supplements and energy drinks), we can use the molecular structure of B12 in Old Tampa Bay to determine if B12 is microbially produced or introduced via wastewater. MICOlab graduate student Lilianna Giuffrida will be processing seawater samples collected by the Florida Fish and Wildlife Conservation Commission Research Institute from Old Tampa Bay throughout the summers of 2025 and 2026. This work is supported by the Tampa Bay Estuary Program TBERF grant.
Explore some preliminary results with this Shiny App!
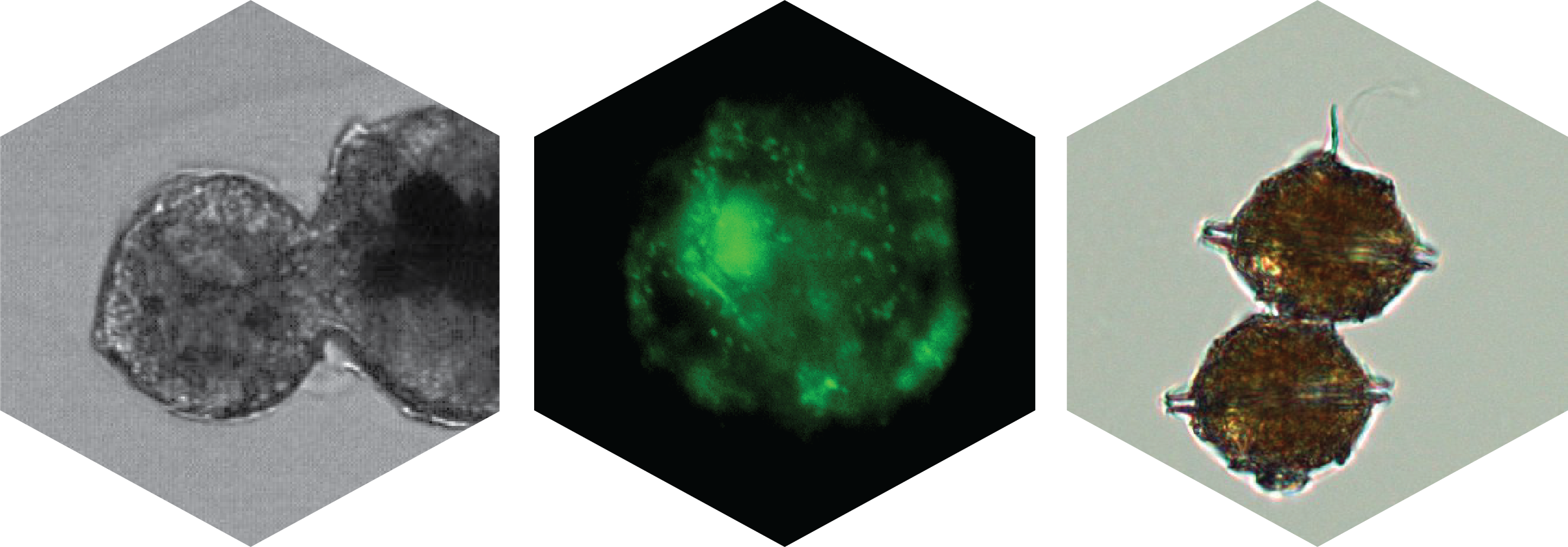
Left: A star is born. Pyrodinium bahamense cell division, caught with an Attune CytPix imaging flow cytometer; Center: Pyrodinium bahamense stained with SYBR green nucleic acid fluorescent dye and imaged with a Zeiss Axioscope 7; Right: A Pyrodinium bahamense doublet imaged with a Zeiss PrimoVert.
The role of mixotrophy and symbiosis in the rise and fall of spring Phaeocystis antarctica blooms along the Western Antarctic Peninsula
The Western Antarctic Peninsula (WAP) region of the Southern Ocean is a highly dynamic and productive ecosystem that is undergoing rapid changes due to global climate change. The spring and summer seasons support high biomass phytoplankton blooms, but the winter is completely devoid of light. Therefore, mixotrophy and symbiosis may be especially important in this ecosystem as a means for photosynthetic organisms to overwinter. Phaeocystis antarctica is an abundant phytoplankton in the Southern Ocean that has a complex multi-morphic life cycle and contributes to myriad mixotrophic and symbiotic arrangements. A key facet of P. antarctica’s life history is its colonial phase, where thousands of cells are embedded in self-secreted mucilaginous, spherical colonies, which make up the majority of Phaeocystis bloom biomass. The colony creates a unique interface for interactions with bacteria and other microeukaryotes: closely related P. globosa colonies host specific microbiomes that can relieve vitamin limitation (Mars Brisbin et al. 2022) and the kelptoplastidic Ross Sea Dinoflagellate uses P. antarctica colonies as a convenient hunting ground by entering colonies and stealing chloroplasts from immobile P. antarctica cells. Phaeocystis species are also the (almost) exclusive photosymbiont of acantharian radiolarians (Mars Brisbin et al. 2018), with acantharians in the Southern Ocean hosting P. antarctica. This project seeks to illuminate how the prevalence of these interactions with P. antarctica in the WAP influnece spring phytoplankton bloom dynamics. Specifically, do bacterial or other interactions prime P. antarctica to outcompete diatoms at the outset of the spring bloom and do interactions with mixotrophic dinoflagellates or other protists (such as acantharians) contribute to the succession from P. antarctica to diatoms in the mid to late spring? MICOlab graduate student Andreas Norlin will be investigating these questions by delving into community-wide gene expression in samples collected during the WAP spring bloom through the Palmer Long Term Ecological Research (LTER) Program.
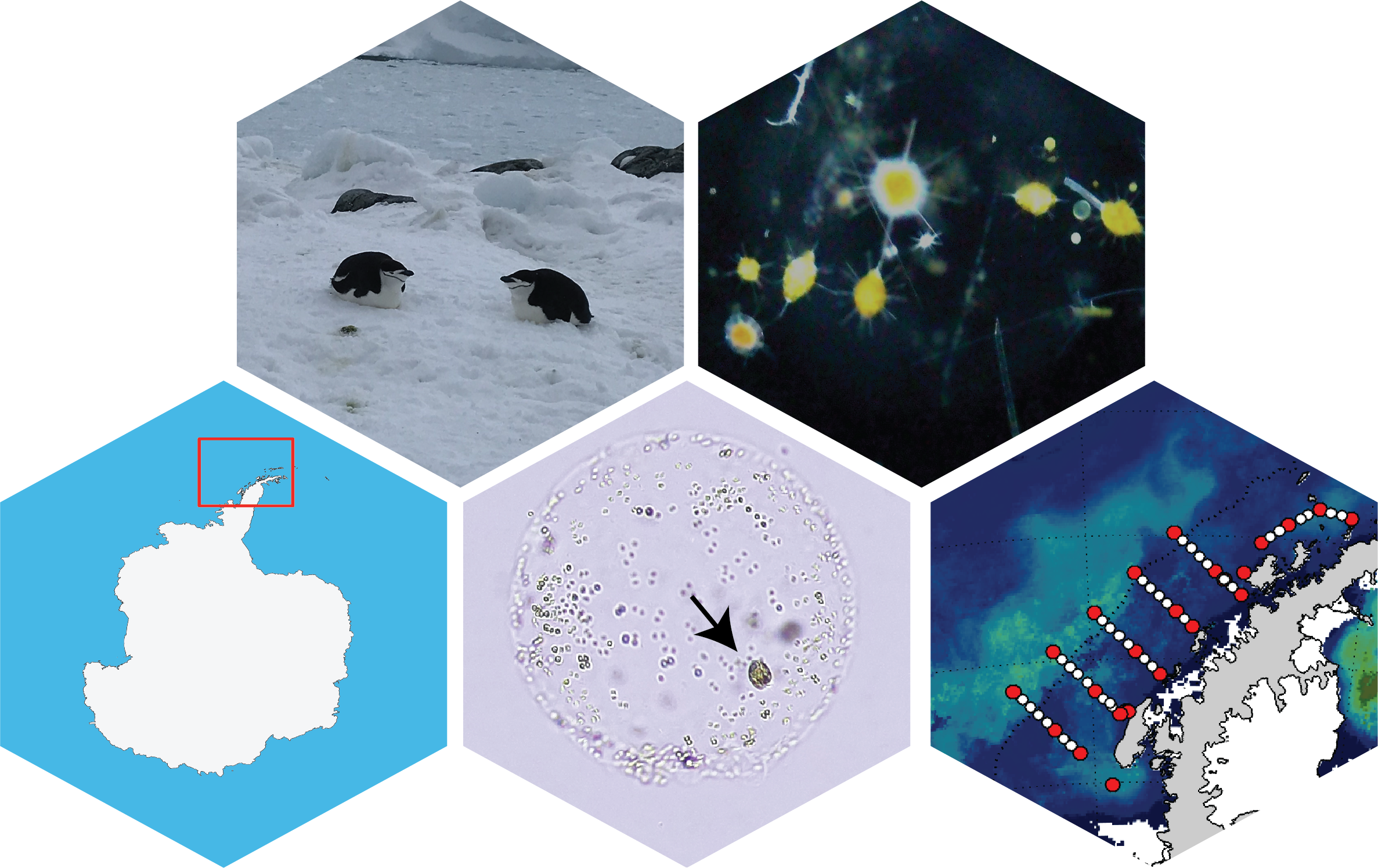 Top left: Chinstrap penguins in the Western Antarctic Peninsula; Top right: An assortment of acantharians with photosynthesic endosymbionts caught during a Phaeocystis bloom off the WAP shelf; Bottom left: Map of Antarctica with the WAP region highlighted in a red square; Bottom middle: A Phaeocystis antarctica colony collected from the spring P. antarctica bloom in the WAP with a kletoplastidic dinoflagellate (arrow) within the colony matrix; Bottom right: Mean spring sea surface chlorophyll concentrations in the WAP in 2021 with the Palmer LTER sampling stations overlaid. Satellite data synthesized by Dr. Jessie Turner, UCONN.
Top left: Chinstrap penguins in the Western Antarctic Peninsula; Top right: An assortment of acantharians with photosynthesic endosymbionts caught during a Phaeocystis bloom off the WAP shelf; Bottom left: Map of Antarctica with the WAP region highlighted in a red square; Bottom middle: A Phaeocystis antarctica colony collected from the spring P. antarctica bloom in the WAP with a kletoplastidic dinoflagellate (arrow) within the colony matrix; Bottom right: Mean spring sea surface chlorophyll concentrations in the WAP in 2021 with the Palmer LTER sampling stations overlaid. Satellite data synthesized by Dr. Jessie Turner, UCONN.
Changing spring bloom composition and phenology in Cape Cod Bay
Phytoplankton dynamics are central to the productivity of marine ecosystems. The species composition of major bloom events determines how much phytoplankton-derived organic carbon is available to higher trophic levels, recycled in the microbial loop, or exported. Hence, changing bloom dynamics have downstream effects on ecosystem functioning and biogeochemical cycling. In the coastal Massachusetts Bay System, including Cape Cod Bay, strong spring diatom blooms have historically supported the higher trophic levels that fuel the vibrant blue economy in the region. Through the Massachusetts Water Resources Authority (MWRA)’s long term monitoring in the region, it has become apparent that major changes in spring bloom timing and composition are underway: the haptophyte Phaeocystis pouchetii has emerged as a dominant contributor to spring bloom biomass, and in 2023, the traditional spring bloom was supplanted by a high-biomass dinoflagellate (Tripos muelleri) bloom that led to early-season benthic hypoxia and anoxia that persisted throughout the summer. In a collaborative effort between Dr. Amy Costa at the Center for Coastal Studies Province Town and Arianna Krinos, Harriet Alexander, and Mak Saito at the Woods Hole Oceanographic Institution, we augmented the last three years of the MWRA time series with whole community meta-omics (i.e., 16S rRNA metabarcoding, metatranscriptomics, and metaproteomics), allowing comprehensive taxonomy and the realized physiological state of different microbes to be directly evaluated across seasonal and annual cycles. Together, these diagnostic ’omics data will shed light on the molecular mechanisms that favor the increasing emergence of Phaeocystis and dinoflagellate blooms in Massachusett’s coastal ecosystems, as well as on how plankton communities respond to marine heatwaves and changing climate patterns.
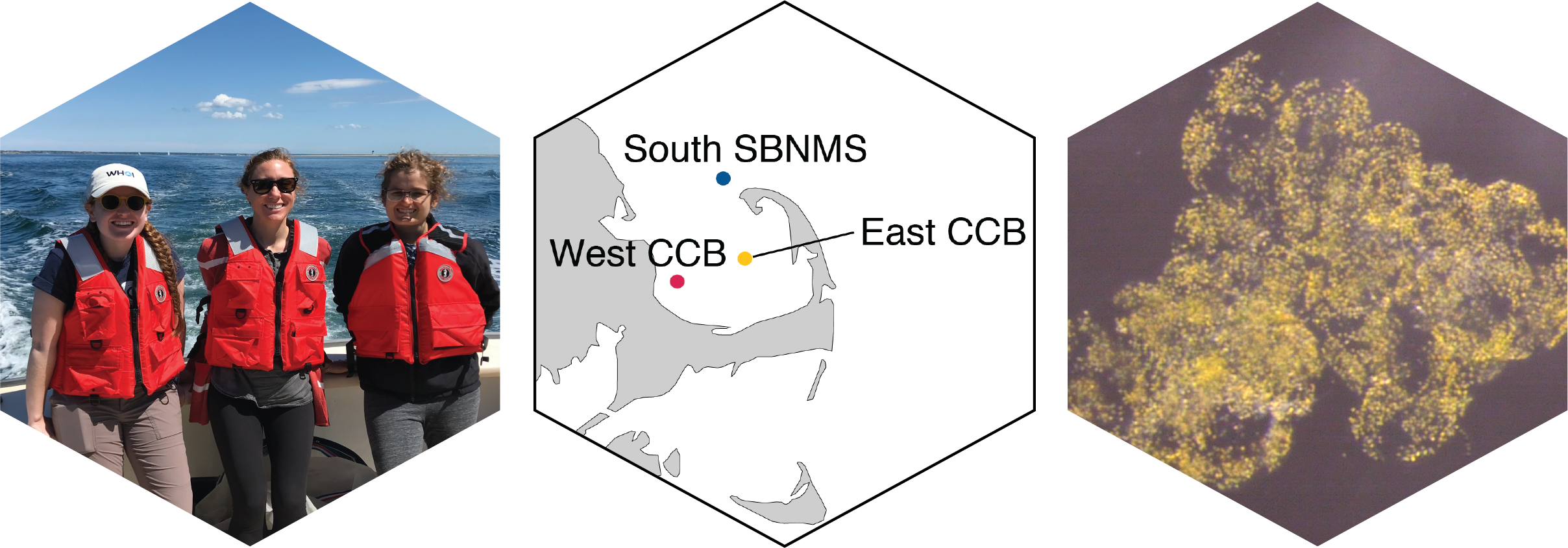
Left: Out sampling in Cape Cod Bay with the Center for Coastal Studies Provincetown; Center: Primary sampling sites for the project in Cape Cod Bay. These sites are a subset of the full MWRA monitoring program. CCB - Cape Cod Bay, SBNMS - Stellwagen Bank National Marine Sanctuary; Right: A Phaeocystis pouchetii colony isolated from Cape Cod Bay water.